Synthesis and Characterization of Bio-Based Polyesters – Bio Based Polyesters FDCA PEF - Arhive
Bio Based Polyesters FDCA PEF Bio Based Polyesters FDCA PEF Bio Based Polyesters FDCA PEF
Synthesis and Characterization of Bio-Based Polyesters:
Poly(2-methyl-1,3-propylene-2,5-furanoate),
Poly(isosorbide-2,5-furanoate), Poly(1,4-cyclohexanedimethylene-2,5-furanoate)
Zoi Terzopoulou 1, Nejib Kasmi 1, Vasilios Tsanaktsis 1, Nikolaos Doulakas 1,
Dimitrios N. Bikiaris 1,*, Dimitris S. Achilias 1 and George Z. Papageorgiou 2,*
1 Laboratory of Polymer Chemistry and Technology, Department of Chemistry,
Aristotle University of Thessaloniki, Thessaloniki GR-541 24, Greece;
terzoe@gmail.com (Z.T.); nejibkasmi@gmail.com (N.K.); vt-vasilis@windowslive.com (V.T.); doulakas@gmail.com (N.D.); axilias@chem.auth.gr (D.S.A.)
2 Department of Chemistry, University of Ioannina, Ioannina GR-54110, Greece
* Correspondence: dbic@chem.auth.gr (D.N.B.); gzpap@cc.uoi.gr (G.Z.P.);
Tel.: +30-231-0997812 (D.N.B.); +30-265-1008354 (G.Z.P.)
Received: 15 June 2017; Accepted: 12 July 2017; Published: 14 July 2017
Abstract: In the present study, three new biobased furanoate polyesters with potential use in food packaging applications, named poly(isosorbide furanoate) (PIsF), poly(methyl-propylene furanoate) (PMePF) and poly(1,4-cyclohexane-dimethylene 2,5-furanoate) (PCHDMF) were synthesized.
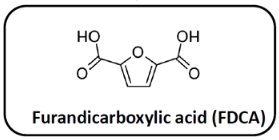 As monomers for the preparation of the polyesters, 2,5-furandicarboxylic acid (FDCA) and diols with irregular or complicated structure were used, including isosorbide (IS), 2-methyl-1,3-propanediol (MPD) and 1,4-cyclohexane-dimethanol (CHDM).
As monomers for the preparation of the polyesters, 2,5-furandicarboxylic acid (FDCA) and diols with irregular or complicated structure were used, including isosorbide (IS), 2-methyl-1,3-propanediol (MPD) and 1,4-cyclohexane-dimethanol (CHDM).
The polymerization process was carried out via melt polycondensation method.
The structural characteristics and thermal behavior of the polymers were studied. The kinetic fragility of the amorphous phase of the polymers was evaluated.
The thermal degradation was studied by means of thermogravimetry and a pyrolysis Py-GC/MS (Pyrolysis-Gas Chromatography/Mass Spectroscopy) system to estimate the degradation mechanism.
1. Introduction
The exploitation of fossil based resources and mainly of petroleum to produce petrochemical polymers has as a result the shrinkage of the above.
Furfural (F) and 5-hydroxymethylfurfural (HMF) can be isolated from carbohydrates, and they constitute precursor compounds of renewable monomers such as 2,5-diformylfuran (DFF) [1], 5-hydroxymethyl-2-furancarboxylic acid (HMFCA) [2] and FDCA [3].
Poly(ethylene furanoate) (PEF) gains ground, against poly(ethylene terephthalate) (PET), especially in food packaging applications, due to renewable sourcing of the monomers and the largely improved barrier, mechanical, and thermal properties compared to PET [4].
